How Do You Know if It a Strong or Weak Acid

Enthalpy of Neutralisation or Heat of Neutralization Chemistry Tutorial
Central Concepts
- Neutralisation, or neutralization, is the name given to the reaction that occurs between an Arrhenius acid and an Arrhenius base.
H+ (aq) + OH- (aq) → H2O(l)
- When an acid is added to an aqueous solution of base, the temperature of the solution increases.
Or, if a base is added to an aqueous solution of an acid, the temperature of the solution increases.
- Free energy (heat) is produced when an acid reacts with a base in a neutralisation reaction.
⚛ Neutralisation reactions are exothermic.
⚛ ΔH for a neutralisation reaction is negative.
- Molar heat of neutralisation (tooth enthalpy of neutralization) is the free energy liberated per mole of h2o formed during a neutralisation reaction.
⚛ ΔHneut is the symbol given to the tooth oestrus of neutralisation.
⚛ ΔHneut is usually given in units of kJ mol-ane
- Heat of neutralisation can be measured in the school laboratory using a styrofoam cup solution calorimter1:
(i) oestrus produced = mass of reaction mixture × specific heat capacity of solution × temperature change
(ii) enthalpy modify for the reaction = -oestrus produced
(3) molar heat of neutralisation = enthalpy change ÷ moles (of water produced)
- Molar heat of neutralisation for reactions between dilute aqueous solutions of stiff acid and stiff base is always the same2, that is,
ΔHneut = -55.90 kJ mol -1
considering no bonds need to exist cleaved, and considering making the H-O bonds in H2O releases energy
(breaking bonds is an endothermic process, making bonds is an exothermic procedure)
- Less than 55.90 kJ mol-ane of energy is released when:
(a) a weak acid neutralises a stiff base of operations
(b) a strong acrid neutralises a weak base
(c) a weak acid neutralises a weak base
because some of the energy is consumed in the procedure of breaking weak acrid bonds or weak base of operations bonds.
Please do non block ads on this website.
No ads = no coin for us = no complimentary stuff for you!
Polstyrene Cup Calorimeter for Rut of Neutralisation Experiment
Expanded polystyrene (polystyrene foam or styrofoam™) cups are often used every bit take-away java cups because the expanded polystyrene is a expert insulator, that is, your coffee stays hot just you don't burn your fingers belongings the cup!
This same insulating belongings can be exploited to make a reasonably skillful calorimeter (a device used to measure out energy, or heat, change during a chemical reaction).
A schematic diagram of a simple polystyrene cream cup calorimeter is shown below:
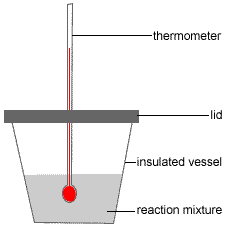
A known amount of a reactant, such as a dilute solution of a base of operations, is placed in the polystyrene loving cup (insulated vessel in the diagram).
The following data for this solution are recorded:concentration: cane = ? mol L-1
volume = Vane mL
A small pigsty is placed in the polystyrene chapeau to allow a thermometer to be pushed through. The fit must be snug enough to hold the thermometer in place, suspended off the bottom of the cup and immersed in the reactant.
It is assumed no heat volition be lost through the lid or the hole in the lid.
The intial temperature of the reactant is measured and recorded.
Ti = ? °C
A known amount of the 2nd reactant, for example a dilute solution of acid, is added to the solution in the cup.
The following information for this second solution are recorded:
concentration: ctwo = ? mol 50-1
book = V2 mL
The thermometer is also used to stir the solution while the reaction is taking identify.
The temperature of the solution in the cup will rise.
The maximum temperature reached is recorded as the final temperature.
Tf = ? °C
For the calculation of estrus of neutralization (enthalpy of neutralisation) nosotros commencement demand to determine:
- full mass, chiliad, of the solution in the cup
First assume additivity of volumes and then that total volume of solution, Vf, is the sum of the book of the two reactants:5f = 5ane + Five2
Then assume that, because all of the solutions are dilute aqueous solutions, the density of each solution and hence the density of the final solution, d, is the aforementioned equally h2o which nosotros will assume is 1.00 chiliad mL-i
density = mass (g) ÷ volume (mL)
Substitute the density of h2o into the equation:
1.00 = mass (g) ÷ Vf
Multiplying both sides of the equation by Vf
Vf × 1.00 =Fivef× mass (yard) ÷Vf
Vf × 1.00 = mass (g) = m - specific rut chapters, Cthou, of the solution
Assume that, because all of the solutions are dilute aqueous solutions, the specific heat capacity of each solution and hence the specific heat capacity of the final solution, Cone thousand, is the same as water which nosotros volition assume is 4.eighteen J yard-i °C-1Cg = 4.xviii J g-1 °C-ane
- change in temperature, ΔT, equally a result of the neutralisation reaction:
ΔT = Tf - Ti °C
Now we tin can calculate the heat released by the neutralisation reaction, q,
q = m × Cg × ΔT
In order to determine the molar oestrus of neutralization (molar enthalpy of neutralisation), we need to determine how many moles of water, n(HiiO(l)), accept been formed as a upshot of the reaction:
H+ (aq) + OH- (aq) → H2O(l)
So, using the stoichiometric ratio (mole ratio), nosotros tin see that:
n(H+ (aq)) = north(OH- (aq)) = north(H2O(50))
Now nosotros can calculate the energy released per mole of water, or the molar enthaply of neutralisation (molar heat of neutralization), ΔHneut:
ΔHneut = -q ÷ due north(H2O(50))
Note that neuralisation is an exothermic reaction, it releases estrus, so ΔHneut must be negative.
Rut of Neutralisation: Strong Monoprotic Acrid + Stiff Monobasic Base of operations
HCl(aq) is a stiff monoprotic acid, it completely dissociates (ionises) in water to produce hydrogen ions (H+ (aq)) and chloride ions (Cl- (aq)):
HCl → H+ (aq) + Cl- (aq)
n(HCl(aq)) : n(H+ (aq)) is 1:1 (that is, monoprotic)
NaOH(aq) is a strong monobasic base of operations, it completely dissociates (ionises) in water to produce sodium ions (Na+ (aq)) and hydroxide ions (OH- (aq)):
NaOH → Na+ (aq) + OH- (aq)
n(NaOH(aq)) : n(OH- (aq)) is 1:1 (that is, monobasic)
A neutralization reaction occurs when HCl(aq) is added to NaOH(aq)
HCl(aq) + NaOH(aq) → H2O(l) + NaCl(aq)
and oestrus energy is given off (the reaction is said to be exothermic)
In an experiment to determine the molar enthalpy of neutralisation, 50.0 mL of 1.0 mol L-1 NaOH(aq) is placed in the styrofoam cup.
The temperature of the NaOH(aq) is recorded.
one.0 mol L-1 HCl(aq) at the aforementioned temperature is added 10.0 mL at a time.
The reaction mixture is stirred betwixt each add-on.
The maximum temperature the solution reached is then recorded.
The results of the experiment are shown in the tabular array below:
| total volume HCl(aq) added (mL) | 0 | 10 | 20 | 30 | 40 | fifty | 60 | 70 |
|---|---|---|---|---|---|---|---|---|
| temperature in calorimeter (°C) | 18.0 | 20.ii | 21.eight | 22.ix | 23.8 | 24.half-dozen | 24.0 | 23.6 |
and the results accept been plotted on the graph shown below:
| temperature °C | HCl(aq) added to NaOH(aq) total volume of HCl(aq) added (mL) |
Initially, the temperature of the reaction mixture in the calorimeter (styrofoam loving cup) increases as HCl(aq) is added.
Energy (oestrus) is being produced by the reaction.
The reaction is exothermic.
Maximum temperature reached is 24.6°C when 50.0 mL of HCl(aq) had been added.
When fifty.0 mL of the acid has been added, all the base of operations has been neutralised.
HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l)
moles HCl(aq) added = moles of NaOH(aq) present in the calorimeter
Adding more acid doesn't increase the temperature in the calorimeter any furtherthree.
We tin can calculate the molar rut of neutralisation for the reaction if we assume:
- the density of each dilute aqueous solution is the same as h2o, 1 g mL-1 at 25°C
so, the mass of solution in grams = volume of solution in mL - the heat chapters of each solution is the same equally for water, 4.18 J°C-aneg-ane
Calculating the molar enthalpy of neutralisation using the data from the experiment:
- Extract the data needed to calculate the tooth oestrus of neutralisation for this reaction:
V(NaOH) = volume of NaOH(aq) in the calorimeter = fifty.0 mL
V(HCl) = book of HCl(aq) added to achieve neutralisation = 50.0 mL
c(NaOH) = concentration of NaOH(aq) = one.0 mol 50-one
c(HCl) = concentration of HCl(aq) = ane.0 mol L-1
Ti = initial temperature of solutions before additions = 18.0°C
Tf = concluding temperature of solution at neutralisation = 24.6°C
d = density of solutions = 1 g mL-1 (assumed)
Cthousand = specific heat capacity of solutions = four.18 J°C-1g-1 (assumed)
q = rut liberated during neutralisation reaction = ? J - Check the units for consistency and catechumen if necessary:
Convert volume of solutions (mL) to mass (m):
density × volume = mass
since density = 1 g mL-1:
1 × volume (mL) = mass (g)
mass(NaOH) = 50.0 grand
mass(HCl) = 50.0 m - Calculate the heat produced during the neutralisation reaction:
oestrus produced = full mass × specific rut capacity × change in temperature
q = mtotal × Cthousand × ΔTmtotal = mass(NaOH) + mass(HCl) = 50.0 + 50.0 = 100.0 g
C1000 = 4.18 J°C-oneyard-one
ΔT = Tf - Ti = 24.half-dozen - 18.0 = 6.half dozen°Cq = 100.0 × 4.eighteen × 6.vi = 2758.viii J
- Calculate the moles of h2o produced:
OH- (aq) + H+ (aq) → H2O(fifty)
1 mol OH- (aq) + one mol H+ (aq) → one mol H2O
moles(HtwoO) = moles(OH- (aq))
moles(OH- (aq)) = concentration (mol L-one) × volume (L)
= 1.0 × 50.0/1000 = 0.050 mol
moles of h2o produced = 0.050 mol - Calculate the heat liberated per mole of h2o produced, ΔHneut :
ΔHneut volition be negative because the reaction is exothermic
ΔHneut = heat liberated per mole of water
= -one × q ÷ moles of water
ΔHneut = -1 × 2758.8 ÷ 0.050
= -55176.0 J mol-iWe tin convert J to kJ by dividing by thousand:
ΔHneut = -55176.0Jmol-1 ÷ 1000J/kJ = 55.2 kJ mol-ane
Enthalpy of Neutralization: Strong Diprotic Acrid and Strong Monobasic Base
The experiment described above is repeated using fifty.0 mL of 1.0 mol L-i sodium hydroxide, a stiff monobasic base, and i.0 mol L-1 sulfuric acid, a stiff diprotic acid, instead of 1.0 mol 50-one muriatic acid, a strong monoprotic acid.
When plotted on a graph as shown beneath, the 2nd experiment'south results look different when compared to the beginning experiment'southward results:
| temperature °C | Graph total volume of acid added (mL) |
Initially, the temperature of the reaction mixture in both experiments increases as acid is added.
Energy (oestrus) is being produced by the reaction. The reaction is exothermic.
Maximum temperature reached for the reaction with H2SOfour(aq) is higher than the maximum temperature reached for the reaction with HCl(aq).
Volume of H2SO(aq) added to reach the maximum temperature is less than the volume of HCl(aq) needed to attain maximum temperature.
For the reaction HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l)
we found that ΔH = -55.2 kJ mol-one (per mole of water formed)
So, H+ (aq) + OH- → HtwoO(l) ΔHneut = -55.ii kJ mol-one
For the reaction HiiSO4(aq) + 2NaOH(aq) → Na2SO4(aq) + 2H2O(fifty)
2H+ (aq) + 2OH- (aq) → 2H2O(l)
so, H+ (aq) + OH- (aq) → HtwoO(l)
We predict that ΔHneut = -55.2 kJ mol-ane
We tin can use the results of the second experiment to calculate the value for the molar heat of neutralisation (ΔHneut), and see if they hold with our prediction.
Summate the molar heat of neutralisation for the reaction:
H2SO4(aq) + 2NaOH(aq) → Na2And sofour(aq) + 2H2O(l)
- Extract the data needed to calculate the molar oestrus of neutralisation for this reaction:
V(NaOH) = book of NaOH(aq) in the calorimeter = 50.0 mL
Five(HiiThen4) = volume of HtwoAnd sofour(aq) added to achieve neutralisation = 25.0 mL
c(NaOH) = concentration of NaOH(aq) = 1.0 mol Fifty-i
c(H2SOfour) = concentration of H2SO4(aq) = 1.0 mol L-1
Ti = initial temperature of solutions earlier additions = xviii.0°C
Tf = final temperature of solution at neutralisation = 26.9°C
d = density of solutions = 1 g mL-1 (causeless)
C1000 = specific heat capacity of solutions = 4.eighteen J°C-1g-1 (assumed)
q = rut liberated during neutralisation reaction = ? J - Bank check the units for consistency and catechumen if necessary:
Catechumen volume of solutions (mL) to mass (one thousand):
density × volume = mass
since density = 1 g mL-1:
1 × volume (mL) = mass (g)
mass(NaOH) = 50.0 g
mass(H2And then4) = 25.0 g - Summate the heat produced during the neutralisation reaction:
estrus produced = total mass × specific heat chapters × change in temperature
q = mtotal × Cg × ΔTmtotal = mass(NaOH) + mass(H2Thenfour() = 50.0 + 25.0 = 75.0 thousand
Cg = iv.18 J°C-1g-1
ΔT = Tf - Ti = 26.9 - 18.0 = 8.9°Cq = 75.0 × 4.18 × 8.ix = 2790.2 J
- Calculate the moles of h2o produced:
OH- (aq) + H+ (aq) → HiiO(l)
ane mol OH- (aq) + one mol H+ (aq) → i mol HtwoO
moles(H2O) = moles(OH- (aq))
moles(OH- (aq)) = concentration (mol L-1) × volume (L)
= 1.0 × l.0/1000
= 0.050 mol
moles of water produced = 0.050 mol - Calculate the heat liberated per mole of water produced, ΔHneut :
ΔHneut will be negative because the reaction is exothermic
ΔHneut = oestrus liberated per mole of h2o
= -one × q ÷ moles of water
ΔHneut = -1 × 2790.ii ÷ 0.050
= -55803 J mol-one (of water produced)
We tin divide J by 1000 to convert this enthalpy change to kJ per mole:
ΔHneut = 55803Jmol-1 ÷ 1000J/kJ
= 55.8 kJ mol-1 (of water produced) - Compare the molar oestrus of neutralisation for:
HCl(aq) + NaOH(aq) → NaCl(aq) + HiiO(l) ΔHneut = -55.ii kJ mol-1 (of water) H2Then4(aq) + 2NaOH(aq) → Na2SO4(aq) + 2HiiO(50) ΔHneut = -55.viii kJ mol-1 (of h2o) There is close agreement betwixt the two values for molar heat of neutralisation, so we can generalise and say that the molar rut of neutralisation for the reaction between a stiff acrid and a strong base is a constant.
Molar Enthalpy of Neutralisation: Weak Acrid + Strong Base
The experiment described above is repeated using 50.0 mL of 1.0 mol L-one sodium hydroxide, a strong monobasic base, and 1.0 mol L-1 hydrogren cyanide (HCN), a weak monoprotic acrid (Granda ≈ 6 × 10-10), instead of one.0 mol Fifty-1 hydrochloric acid, a potent monoprotic acid.
The results of these experiments is shown in the graph below:
| temperature °C | Graph full volume of acid added (mL) |
Initially, the temperature of the reaction mixture in both experiments increases equally acid is added.
Energy (rut) is being produced by the reaction. The reaction is exothermic.
Maximum temperature reached for the reaction with HCN(aq) is much less than the maximum temperature reached for the reaction with HCl(aq).
Volume of HCN(aq) added to reach the maximum temperature is the same as the volume of HCl(aq) needed to achieve maximum temperature (both volumes are 50.0 mL).
For the reaction HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(fifty)
nosotros found that ΔH = -55.ii kJ mol-1 (per mole of water formed)
And then, H+ (aq) + OH- → H2O(l) ΔHneut = -55.ii kJ mol-1
For the reaction HCN(aq) + NaOH(aq) → NaCN(aq) + H2O(l)
Will ΔH for this reaction be the same (-55.2 kJ mol-1) ?
We can apply the results of the HCN experiment to calculate the value for the tooth rut of neutralisation (ΔHneut), and run across.
- Extract the information needed to calculate the tooth heat of neutralisation for this reaction:
5(NaOH) = volume of NaOH(aq) in the calorimeter = 50.0 mL
V(HCN) = book of HCN(aq) added to attain neutralisation = l.0 mL
c(NaOH) = concentration of NaOH(aq) = 1.0 mol L-1
c(HCN) = concentration of HCN(aq) = one.0 mol L-1
Ti = initial temperature of solutions earlier additions = eighteen.0°C
Tf = concluding temperature of solution at neutralisation = xix.2°C
d = density of solutions = 1 thou mL-1 (assumed)
Cone thousand = specific estrus chapters of solutions = iv.18 J°C-11000-i (assumed)
q = estrus liberated during neutralisation reaction = ? J - Check the units for consistency and catechumen if necessary:
Convert volume of solutions (mL) to mass (one thousand):
density × volume = mass
since density = i m mL-ane (assumed):
1 × volume (mL) = mass (chiliad)
mass(NaOH) = 50.0 thousand
mass(HCN) = l.0 k - Calculate the estrus produced during the neutralisation reaction:
heat produced = total mass × specific heat capacity × modify in temperature
q = mfull × Cg × ΔTmtotal = mass(NaOH) + mass(HCN) = 50.0 + 50.0 = 100.0 g
Cg = 4.18 J°C-1g-1
ΔT = Tf - Ti = 19.two - 18.0 = 1.ii°Cq = 100.0 × iv.18 × 1.2 = 501.6 J
- Calculate the moles of h2o produced:
NaOH(aq) + HCN(aq) → NaCN(aq) + H2O(l)
moles(H2O) = moles(NaOH)
moles(NaOH(aq)) = concentration (mol L-1) × book (L)
= 1.0 × 50.0/1000 = 0.050 mol
moles of water produced = 0.050 mol - Calculate the estrus liberated per mole of water produced, ΔHneut :
ΔHneut volition be negative considering the reaction is exothermic
ΔHneut = heat liberated per mole of water
= -1 × q ÷ moles of water
ΔHneut = -1 × 501.six ÷ 0.050
= -10032 J mol-1We tin can catechumen J to kJ past dividing by 1000:
ΔHneut = -10032Jmol-1 ÷ 1000J/kJ
= -x.0 kJ mol-1 - Compare the molar rut of neutralisation for the neuralisation of NaOH(aq) past both HCl(aq) and HCN(aq):
HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(fifty) ΔHneut = -55.ii kJ mol-1 (of h2o) HCN(aq) + NaOH(aq) → NaCN(aq) + H2O(l) ΔHneut = -10.0 kJ mol-1 (of h2o) The heat released per mole of water for the hydrogen cyanide reaction is much less than the heat released per mole of water for the hydrochloric acid reaction.
The departure in molar heats of neutralisation is due to the blazon of reaction taking place:
- Strong Acid + Potent Base Reaction:
⚛ Potent base of operations, NaOH, fully dissociates in water. The reacting species is OH- (aq)
⚛ Strong acid, HCl, fully dissociates in water. The reacting species is H+ (aq)
⚛ The reaction is therefore an Arrhenius neutralisation reaction:
H+ (aq) + OH- (aq) → HiiO(l)
⚛ No bonds need to broken in the stiff acid or strong base, no free energy is lost in breaking bonds.
⚛ Energy is produced when the H-O bonds class in the HtwoO product.
- Weak Acrid + Strong Base
⚛ Strong base, NaOH, fully dissociates in water. The reacting species is OH- (aq)
⚛ Weak acid, HCN, only partially dissociates in water.
Most of the acrid species in solution are undissociated HCN molecules.⚛ The reaction is therefore a Brønsted-Lowry proton transfer reaction:
HCN(aq) + OH- (aq) → CN- (aq) + H2O(fifty)
⚛ In order for this reaction to occur, H-C bonds in HCN molecules must be broken before H2O molecules can be produced.
Breaking covalent bonds requires energy.⚛ And then, even though we might expect the aforementioned amount of energy to exist produced in both the HCl and HCN reactions because both reactions are producing the same number of moles of HiiO, nosotros encounter that energy will be consumed in breaking bonds in HCN so the amount of free energy produced overall will be less than that for the HCl + NaOH reaction.
The heat liberated per mole when a weak acrid neutralises a strong base of operations is less than the corporeality of rut liberated per mole when a potent acid neutralises a strong base.
Footnotes
1. A better method for measuring heat of neutralization is to use an adiabatic calorimeter fitted with an electrical heater. A desciption of this type of calorimeter tin be establish in the calorimetry tutorial.
2. You will notice slightly dissimilar values quoted for molar heat of neutralisation generally because the neutralisation reaction is dependent on the temperature at which the reaction occurs. In general the values you see quoted will exist between 55 kJ mol-1 and 58 kJ mol-one and refer to reactions that have identify at ambience temperatures in a laboratory.
three. The temperature is likely to decrease as more acid is added because the heat that was generated by the completed reaction is being prodigal in a greater mass of solution.
Source: https://www.ausetute.com.au/heatneutral.html
0 Response to "How Do You Know if It a Strong or Weak Acid"
Post a Comment